Neurodegenerative Disease Biomarker Discovery Accelerated
Neurodegenerative diseases are tricky to manage because they are often difficult to diagnose. An incorrect diagnosis can lead to costly and unnecessary treatments, worsening of the disease, and family stress and frustration. For dementia, such as Alzheimer’s, diagnosis is determined through a “rule-out” process where the patient endures several layers of testing – physical exams, brain scans, memory tests and family interviews. This process can take months. Similarly, a diagnosis of Parkinson’s disease can take just as long with behavioral tests, brain scans, painful spinal tap, and evaluation of family history taking considerable time.

Despite the distinct symptoms characterizing each neurodegenerative disease, they share a common challenge: the considerable difficulty in achieving early diagnosis.
To expedite diagnoses and accelerate intervention, researchers are exploring new innovative methods to identify neurodegenerative diseases before symptoms reach critical stages. One promising, non-invasive approach involves the immune system. The humoral immune system continuously surveys the body for foreign or out-of-place substances, most often aberrantly positioned proteins. These proteins are not only from invading organisms such as parasites or bacteria, but they can also be produced from an autoantigen, for example the aggregate forming 43 kd transactive response DNA binding protein (TDP-43) found in the ALS and Alzheimer’s brains stimulates anti-TDP43 autoantibody expression (Conti et al., 2021). Detecting these autoantibodies in blood serum can be achieved through methods like ELISA or protein arrays. However, ELISA, while effective, presents limitations in discovering new biomarkers due to inherent background noise and low throughput.
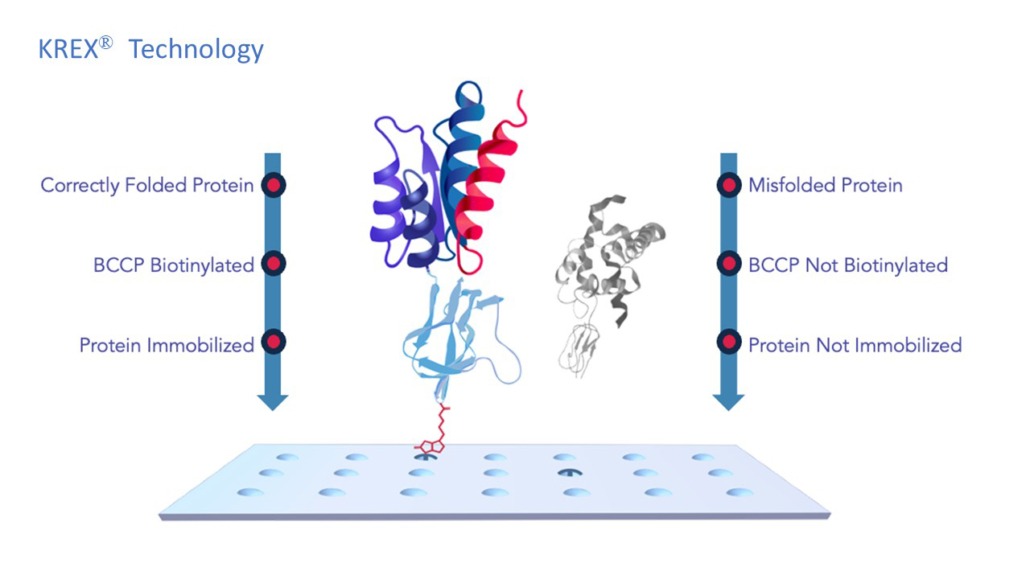
Protein microarrays offer a high-throughput method to analyze numerous potential autoantibodies from patient sera quickly and efficiently. The Sengenics i-Ome Discovery microarray consists of more than 1800 carefully curated protein antigens, with ~25% of the array proteins related to over 3000 neuronal related diseases.
The arrays are manufactured with Sengenics’s patented KREX® protein folding technology that ensures proper protein folding and consistent protein orientation on the array across every printing. KREX® establishes these arrays as exceptionally dependable and reproducible tools for proteomic research.
This technology was used to gain a better understanding of age-related immune dysfunction that may participate in the pathogenesis of dementia. Examination of autoantibody profiles from serum samples of patients with mild cognitive impairment and dementia revealed distinct autoantibody signatures for each condition compared to controls. Interestingly, each disease shared dysregulation of 5 specific autoantibodies including antibodies to CAMK2A, CKS1B, ETS2, MAP4 and NUDT2.
An advantage of these arrays, sometimes overlooked, regards the bidirectionality of the results. Some autoantibodies were upregulated, and some were down regulated in diseased conditions compared to normal individuals. This indicated a potentially protective role of some autoantibodies in age-related loss of cognitive function while also illuminating potentially valuable targets for treating dementia. Of note, antibodies against CAMK2A were downregulated in both mild cognitive impairment and dementia patients. CAMK2A dysregulation has been noted in Alzheimer’s (Ehtewish et al., 2023). This kinase participates in synaptic plasticity and cardiac function. CAMK2A inhibitors such as KN-93 are currently under investigation in in vitro and animal models.
Recent advancements over the past two decades, including KREX® protein folding technology in protein microarrays, have yielded progress in the discovery of novel biomarkers for detection of neurodegenerative diseases. Autoantigen identification from patient sera not only offers an early detection of disease, but also provides clues to the protein pathophysiology of disease that can be exploited to develop new and innovative therapeutic interventions.
Have a look at the publications page to learn more:
Sengenics | Precision Medicine Company | Publications
References
Conti, E., et al. (2021). Serum naturally occurring anti-TDP-43 auto-antibodies are increased in amyotrophic lateral sclerosis. Sci Rep, 11(1), 1978. https://doi.org/10.1038/s41598-021-81599-5
Ehtewish, H., et. al. H. (2023). Profiling the autoantibody repertoire reveals autoantibodies associated with mild cognitive impairment and dementia [Original Research]. Frontiers in Neurology, 14. https://doi.org/10.3389/fneur.2023.1256745